New test kit that detects Covid-19 and seasonal flu approved by HSA
A new multi-virus test kit that is able to detect both Covid-19 and the seasonal flu has obtained provisional authorisation from Singapore's Health Sciences Authority.
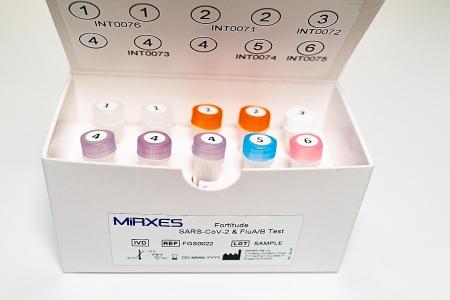
The Fortitude Sars-CoV-2 and Flu A/B test kit was jointly developed by the Agency for Science, Technology and Research, Tan Tock Seng Hospital and local molecular diagnostic company MiRXES.
Building on the existing Fortitude Kit that tests only for Covid-19, this new kit has additional polymerase chain reaction (PCR) primers - short genetic sequences - designed to detect influenza A and B.
The test uses PCR to look for the viruses that cause Covid-19 or the flu and will give two readouts, showing if the patient has either.
Since February last year, more than five million Fortitude Kit tests have been sold locally and globally.
The kits have been deployed in 13 local hospitals and labs and exported to more than 40 countries, including the United States.
Sales of the new kit have just been launched, after it obtained the regulatory approval on Jan 8.
Patients with Covid-19 or flu exhibit a set of similar symptoms, making it difficult for healthcare professionals to differentiate between the two. This new kit will allow for a more accurate and timely diagnosis.
MiRXES has also obtained the Conformite Europeenne mark approval, a European certification, for the new kit.
The company is applying for emergency use authorisation from the US Food and Drug Administration, and for approval from Japan's Pharmaceuticals and Medical Devices Agency. - THE STRAITS TIMES
Get The New Paper on your phone with the free TNP app. Download from the Apple App Store or Google Play Store now